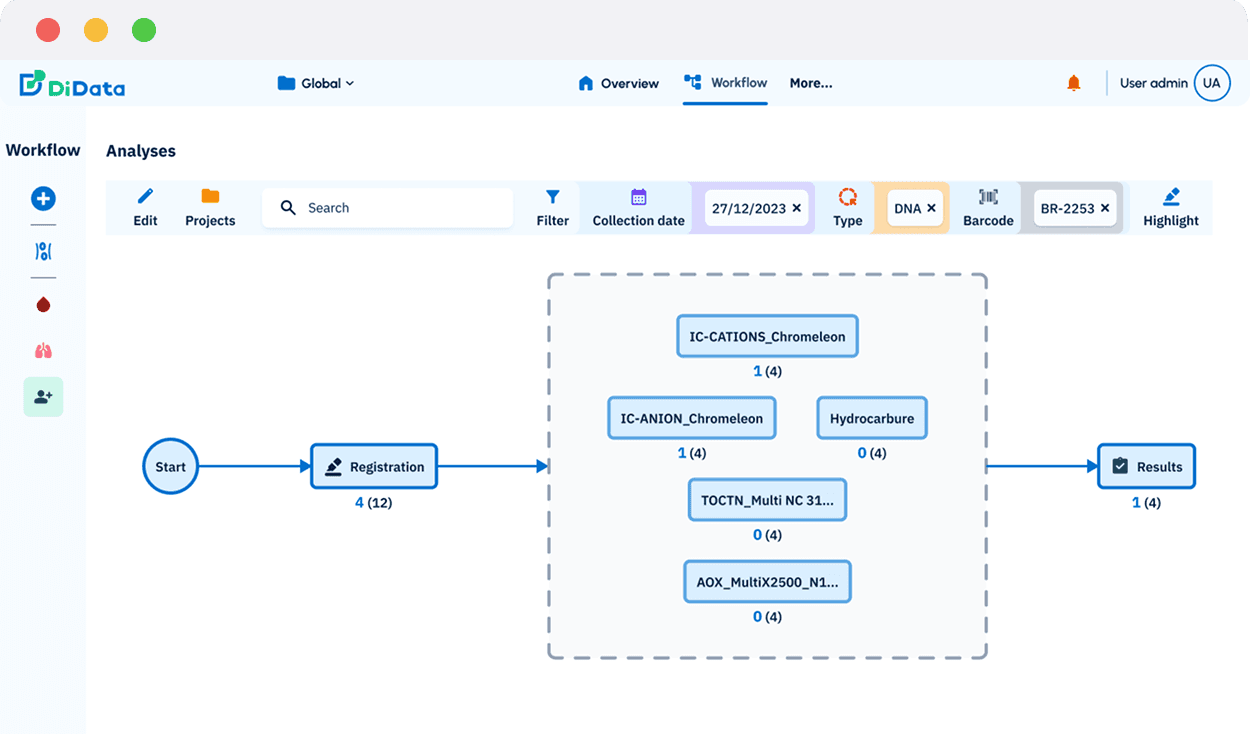
“With DiData, we’ve gained full control and confidence in our biobank operations. Data safety is guaranteed thanks to local hosting on our own servers and automatic backups. We can now manage complex study designs through a single, centralized system, no more fragmented sampling schemes. The platform’s flexibility also makes a big difference: admins can easily reprint or correct labels during hospital collections, keeping everything accurate and consistent.”
Biobank Operations Lead





.png&w=1920&q=75)



.png&w=3840&q=75)